|
|
Anticoagulant Phospholipase A2 (PLA2) Enzyme
|
Some snake venom PLA2 enzymes inhibit blood coagulation. Based on their anticoagulant properties they are classified into strongly, weakly and non-anticoagulant enzymes. Since phospholipids play a crucial role in the formation of several coagulation complexes, intuitively one might anticipate that the destruction of phospholipid surface would be the primary mechanism to account for anticoagulant effects of PLA2 enzymes. However, the role of enzymatic activity in anticoagulant effects was controversial and the mechanism was unclear. We solved the mechanism of anticoagulant effects of strongly anticoagulant PLA2 enzyme from Naja nigricollis venom.
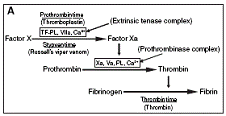 Dissection approach – We designed a simple approach to determine the particular step at which an anticoagulant interferes in the extrinsic pathway. This “Dissection Approach” is rapid, cost effective and employs three commonly used clotting assays, namely prothrombin time, Stypven time and thrombin time. It is based on the simple principle that initiating the cascade "upstream" from the inhibited step will result in elevated clotting times, while initiating the cascade "downstream" from the inhibited step will not affect the clotting time. Dissection approach – We designed a simple approach to determine the particular step at which an anticoagulant interferes in the extrinsic pathway. This “Dissection Approach” is rapid, cost effective and employs three commonly used clotting assays, namely prothrombin time, Stypven time and thrombin time. It is based on the simple principle that initiating the cascade "upstream" from the inhibited step will result in elevated clotting times, while initiating the cascade "downstream" from the inhibited step will not affect the clotting time.
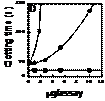 Localization of site of inhibition – Using the dissection approach as well as the inhibition studies of specific reconstituted complexes, we showed that the extrinsic tenase (tissue factor–Factor VIIa) complex is inhibited by all three anticoagulant PLA2 enzymes (CM-I, CM-II and CM-IV) from N. nigricollis venom, whereas the prothrombinase complex is inhibited only by CM-IV. Thus the strongly anticoagulant enzyme CM-IV inhibits both the extrinsic tenase and prothrombinase complexes, creating two ‘bottlenecks’ in the coagulation cascade, whereas the weakly anticoagulant enzymes CM-I and CM-II only inhibit the extrinsic tenase complex, and create only a single bottleneck. Localization of site of inhibition – Using the dissection approach as well as the inhibition studies of specific reconstituted complexes, we showed that the extrinsic tenase (tissue factor–Factor VIIa) complex is inhibited by all three anticoagulant PLA2 enzymes (CM-I, CM-II and CM-IV) from N. nigricollis venom, whereas the prothrombinase complex is inhibited only by CM-IV. Thus the strongly anticoagulant enzyme CM-IV inhibits both the extrinsic tenase and prothrombinase complexes, creating two ‘bottlenecks’ in the coagulation cascade, whereas the weakly anticoagulant enzymes CM-I and CM-II only inhibit the extrinsic tenase complex, and create only a single bottleneck.
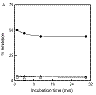 Mechanism of inhibition of the prothrombinase complex – The strongly anticoagulant PLA2 (CM-IV) inhibits the prothrombinase complex via a non-enzymatic mechanism. The inhibition of the reconstituted complex does not increase with an increase in incubation time with CM-IV. In contrast, weakly anticoagulant CM-I and CM-II fail to inhibit the prothrombinase complex even after 30 min incubation. Despite the complete hydrolysis of phospholipids by CM-I and CM-II, thrombin formation is not significantly reduced. Thus the inhibition of the prothrombinase complex is independent of phospholipid hydrolysis. Alkylation of the active-site residue His48 in CM-IV results in the loss of enzymatic activity, but it retains more than 60%of the inhibition of the prothrombinase complex. Furthermore, CM-IV inhibits thrombin formation more strongly in the absence of phospholipids than in their presence. Studies of inhibition kinetics indicate that CM-I Mechanism of inhibition of the prothrombinase complex – The strongly anticoagulant PLA2 (CM-IV) inhibits the prothrombinase complex via a non-enzymatic mechanism. The inhibition of the reconstituted complex does not increase with an increase in incubation time with CM-IV. In contrast, weakly anticoagulant CM-I and CM-II fail to inhibit the prothrombinase complex even after 30 min incubation. Despite the complete hydrolysis of phospholipids by CM-I and CM-II, thrombin formation is not significantly reduced. Thus the inhibition of the prothrombinase complex is independent of phospholipid hydrolysis. Alkylation of the active-site residue His48 in CM-IV results in the loss of enzymatic activity, but it retains more than 60%of the inhibition of the prothrombinase complex. Furthermore, CM-IV inhibits thrombin formation more strongly in the absence of phospholipids than in their presence. Studies of inhibition kinetics indicate that CM-I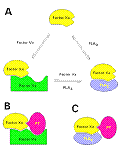 V is a non-competitive inhibitor. Thus CM-IV neither competes with prothrombin to bind to the active site of the prothrombinase complex nor binds to prothrombin. However, CM-IV competes with FVa ( Factor Va) and interferes with complex formation. The addition of increasing amounts of FVa to the mixture reverses the inhibition, suggesting that CM-IV may compete with FVa for binding to FXa (Factor Xa). Direct binding studies using isothermal calorimetry showed that CM-IV forms a 1:1 complex with FXa (Kd 500 nM) and blocks the formation of the prothrombinase complex. Thus the strongly anticoagulant PLA2 enzyme CM-IV inhibits the key step in blood coagulation by a novel non-enzymatic mechanism, and FXa is the target protein for the anticoagulant activity of this enzyme. V is a non-competitive inhibitor. Thus CM-IV neither competes with prothrombin to bind to the active site of the prothrombinase complex nor binds to prothrombin. However, CM-IV competes with FVa ( Factor Va) and interferes with complex formation. The addition of increasing amounts of FVa to the mixture reverses the inhibition, suggesting that CM-IV may compete with FVa for binding to FXa (Factor Xa). Direct binding studies using isothermal calorimetry showed that CM-IV forms a 1:1 complex with FXa (Kd 500 nM) and blocks the formation of the prothrombinase complex. Thus the strongly anticoagulant PLA2 enzyme CM-IV inhibits the key step in blood coagulation by a novel non-enzymatic mechanism, and FXa is the target protein for the anticoagulant activity of this enzyme.
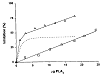 Mechanism of inhibition of the extrinsic tenase complex – All three PLA2 enzymes from N. nigricollis venom inhibit the extrinsic tenase (TF–FVIIa) complex. Interestingly, the strongly anticoagulant enzyme CM-IV inhibits the tenase by both enzymatic and non-enzymatic mechanisms. On the other hand, weakly anticoagulant CM-I and CM-II inhibit the complex mostly by their enzymatic activity. Mechanism of inhibition of the extrinsic tenase complex – All three PLA2 enzymes from N. nigricollis venom inhibit the extrinsic tenase (TF–FVIIa) complex. Interestingly, the strongly anticoagulant enzyme CM-IV inhibits the tenase by both enzymatic and non-enzymatic mechanisms. On the other hand, weakly anticoagulant CM-I and CM-II inhibit the complex mostly by their enzymatic activity.
Key Publications
|
|